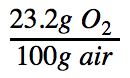Research and Calculations:
How many oxygen (O2) molecules are in one cubic meter of dry air at 0°C and 101.325 kPa?
|
In order to answer this question, I first had to find the mass of one cubic meter of dry air at 0°C and 101.325 kPa. After doing some research, I was able to find that the density of air at 0°C and 101.325 kPa is 1.2929 kg/m^3, as shown in Table 2: Physical Properties of Standard Dry Air below. I converted kilograms (kg) to grams (g) by multiplying by 1000, and determining that there are 1292.9 grams of air in 1 cubic meter of air at 0°C and 101.325 kPa. Using this information, I created the following conversion factor:
|
|
Next, I found the standard composition of dry air and the percentage of oxygen in the air by mass. Since weight and mass are proportional to each other, we can use the terms interchangeably in the context of percentages. So while Table 1 reads "% by weight", we can interpret it as precent by mass. It is important that we use percent by mass, instead of percent by volume, because our other calculations are in mass (grams) and they must be this way so we are able to make the necessary conversions to answer the final question. According to Table 1: Standard Composition of Dry Air on the left, 23.20% of the mass of dry air is oxygen. From basic understanding of percentages and by applying the unit of grams, this can be rewritten as 23.2 grams of O2 per 100 grams of air, as represented by the following conversion factor:
|
|
Next, I had to find the molar mass of oxygen (O2). According to the periodic table of elements, oxygen (O) has an average atomic mass of 15.9994 amu, which I rounded up to 16 amu. Since there are two oxygen atoms in each O2 molecule, I made the calculation 16(2)=32 to find the molecular mass, which is the mass of one molecule of O2. The molecular mass (measured in atomic mass units [amu]) is equal to the molar mass (measured in grams per mole [g/mol]) so by converting from atomic mass units to grams, we have found the molar mass of O2, which is approximately 32 grams per mole. Using this information, I created the following conversion factor:
|
|
Finally, I had to convert from the number of moles to molecules in order find the answer to my research question. To do this, I used Avogadro's number, 6.022*10^23, which is defined as the number of units (particles, molecules, atoms, ions, etc.) in one mole of any substance. In this case, there are 6.022*10^23 molecules of O2 in 1 mole of O2, as represented by the following conversion factor:
|
To find the final answer to my research question, the number of oxygen (O2) molecules in one cubic meter of dry air at 0°C and 101.325 kPa, I multiplied all of my conversion factors together. All of the irrelevant units canceled out and left the final answer, as shown in the calculations below:
There are 5.6447*10^24 molecules of oxygen (O2) in one cubic meter of dry air at 0°C and 101.325 kPa.
There are 5.6447*10^24 molecules of oxygen (O2) in one cubic meter of dry air at 0°C and 101.325 kPa.